Sequential revenue growth of more than 50% compared to Q2 2021
Year-to-date product and service revenue growth of 36% compared to the same period of 2020
BOSTON, MA – November 4, 2021 – 908 Devices Inc. (Nasdaq: MASS), a pioneer of purpose-built handheld and desktop mass spec devices for chemical and biomolecular analysis, today reported financial results for the quarter ended September 30, 2021.
Recent Highlights
- Total revenue for Q3 2021 increased 52% compared to Q2 2021 and 107% compared to the prior year period
- Expanded installed base to 1,714 devices with 184 devices placed during the third quarter, as compared to 103 devices in the second quarter
- Launched an Oligonucleotides analysis kit for the ZipChip device, providing a simple, sensitive method for rapid analysis addressing an additional class of therapeutics
- Partnered with U.S. federal and state officials, including the Ohio Attorney General’s office, to deploy the MX908 handheld device in the field for trace identification of counterfeit pharmaceuticals and other priority drugs
- Hosted inaugural integrated user meeting covering the full breadth of our products and platforms with talks from participants including Amgen, Merck, AstraZeneca Pharmaceuticals, Dana Farber Cancer Institute, and the United States Department of Agriculture, among others
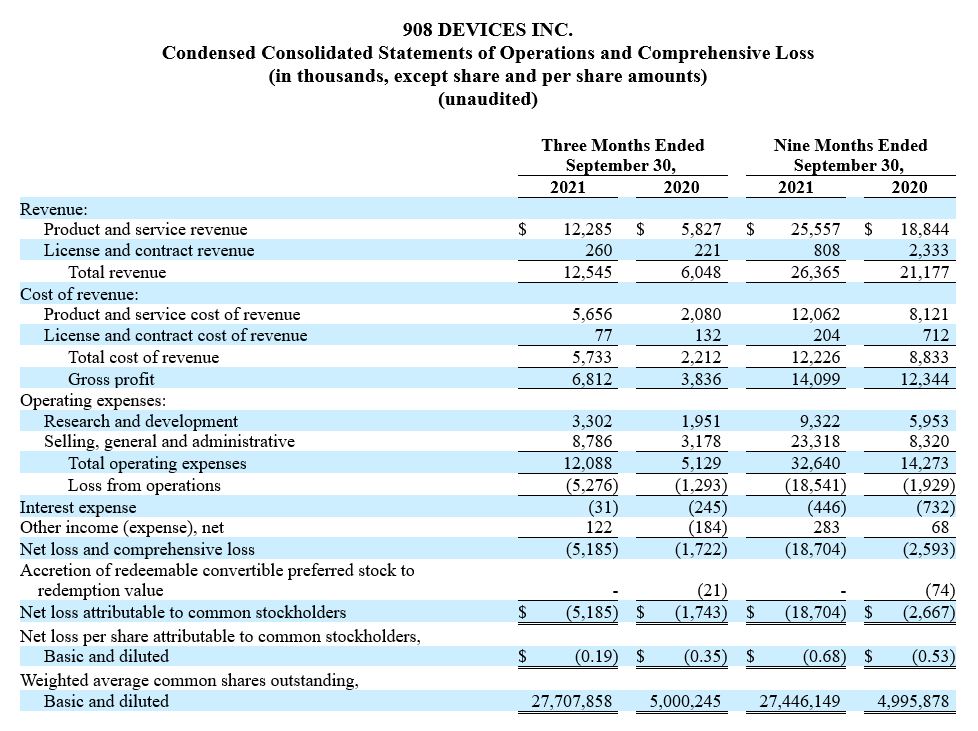
Third Quarter 2021 Financial Results
Revenue was $12.5 million for the three months ended September 30, 2021, a 52% increase over the three months ended June 30, 2021, and a 107% increase compared to the corresponding prior year period. An increase in handheld shipments, including initial shipments to the US Army under the multi-year purchase order, was primarily responsible for the quarter over quarter and the year over year increases. Desktop revenue grew 72% year over year. More than 40% of the desktop units sold during the quarter were placed at top 20 pharmaceutical companies.
The install base grew to 1,714 placements with 184 devices placed in Q3 2021. The increase in devices was driven primarily by growing adoption of our handheld devices.
Gross profit was $6.8 million for the third quarter of 2021, compared to $3.8 million for the corresponding prior year period. Product and service gross margin was 54%, as compared to 64% for the corresponding prior year period. The decline in product and service gross margin was primarily driven by increased investments in 2021 related to manufacturing and service personnel combined with lighter spend during the third quarter of 2020 related to impacts of the COVID-19 pandemic.
Operating expenses were $12.1 million for the third quarter of 2021, compared to $5.1 million for the corresponding prior year period. This increase was driven by headcount expansion across our business, primarily focused on our commercial organization, and increased expenses related to operating as a public company.
Net loss was $5.2 million for the third quarter of 2021, compared to a net loss of $1.7 million for the corresponding prior year period. Net loss per share was $0.19 for the third quarter of 2021, compared to a net loss per share of $0.34 for the corresponding prior year period.
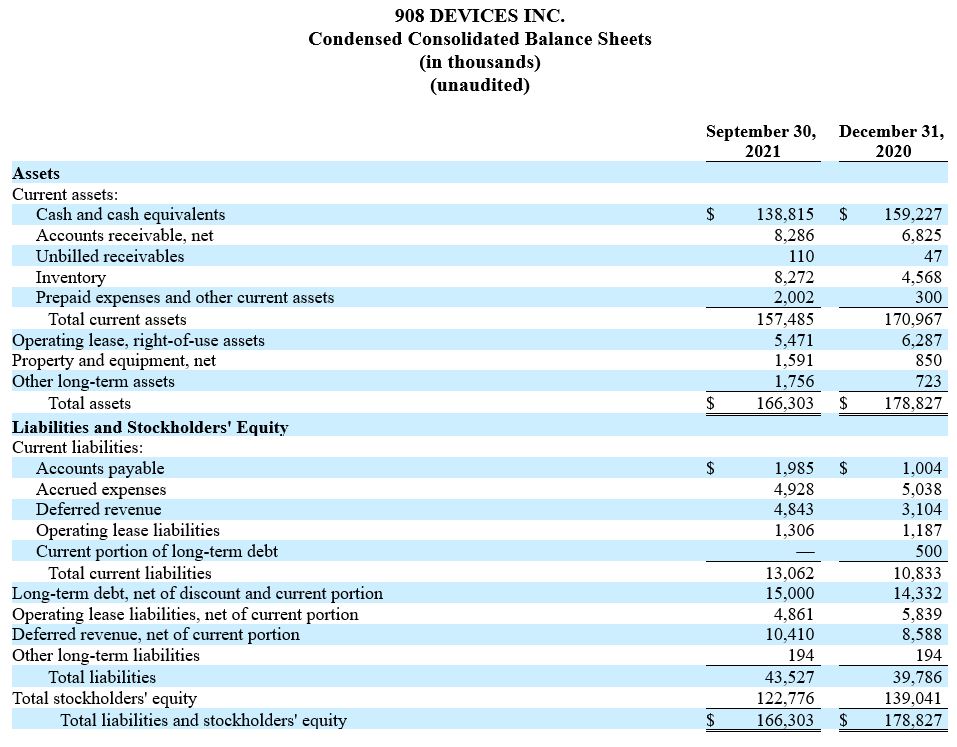
Cash and cash equivalents were $139 million as of September 30, 2021. In addition, the Company has approximately $15 million of debt outstanding.
2021 Guidance
908 Devices now expects full year 2021 revenue to be in the range of $41 million to $43 million, representing 52% to 60% growth over full year 2020. This compares to the previously expected revenue range of $40 million to $43 million.
Webcast Information
908 Devices will host a conference call to discuss the third quarter 2021 financial results before market open on Thursday, November 4, 2021 at 5:30 am Pacific Time / 8:30 am Eastern Time. A webcast of the conference call can be accessed at https://ir.908devices.com/news-events/events. The webcast will be archived and available for replay for at least 90 days after the event.
About 908 Devices
908 Devices is democratizing laboratory mass spectrometry with its simple handheld and desktop devices, addressing critical-to-life applications. The Company’s devices are used at the point-of-need to interrogate unknown and invisible materials and provide quick, actionable answers to directly address some of the most critical problems in life sciences research, bioprocessing, pharma / biopharma, forensics and adjacent markets. The Company is headquartered in the heart of Boston, where it designs and manufactures innovative products that bring together the power of mass spectrometry, microfluidic separations, software automation, and machine learning.
Forward Looking Statements
This press release includes “forward looking statements,” including statements relating to the Company’s future revenue and growth. Forward-looking statements involve known and unknown risks, uncertainties and assumptions which may cause actual results to differ materially from any results expressed or implied by any forward-looking statement, including the risks outlined under “Risk Factors” and elsewhere in the Company’s filings with the Securities and Exchange Commission which are available on the SEC’s website at www.sec.gov. Additional information will be made available in our annual and quarterly reports and other filings that we make from time to time with the SEC. Although the Company believes that the expectations reflected in its forward-looking statements are reasonable, it cannot guarantee future results. The Company has no obligation, and does not undertake any obligation, to update or revise any forward-looking statement made in this press release to reflect changes since the date of this press release, except as may be required by law.